
In a placebo-controlled, double-blind, parallel-group comparison of single oral doses of lorazepam (2 mg) and flunitrazepam (1.2 mg), 36 young, healthy subjects completed a test battery before and after treatment including classic behavioral tests and visual and auditory event-related potentials. These results suggested that lorazepam can cause significant impairment of driving and psychomotor abilities, independent of the blood concentration. 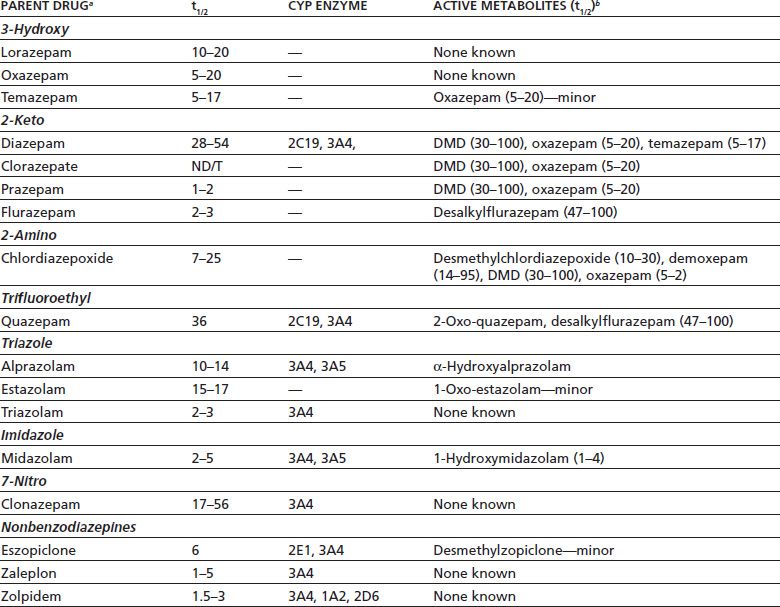
In ten of the other cases (in which no drugs other than lorazepam were present) Drug Recognition Expert reports were obtained, containing details of events surrounding arrests and performances on field sobriety tests lorazepam concentrations in these cases averaged 50 ng/ml. In 23 cases lorazepam was the only drug detected, at a mean blood concentration of 51 ng/ml. The mean whole lorazepam blood concentration in these drivers (n = 170, 56% male, mean age 40 years) was 48 ng/ml, but 86% of drivers tested positive for other drugs, which may have contributed to their impairment. All positive lorazepam drug-impaired driving cases submitted to the Washington State Toxicology Laboratory between January 1998 and December 2003 were reviewed. Common adverse effects include sedation, dizziness, weakness, unsteadiness, and disorientation, and lorazepam can cause significant impairment of driving ability. Lorazepam is often used to manage anxiety, presurgically, and as a sedative. IM and oral administration of lorazepam in elderly persons, as in the case of young individuals, leads to rapid and nearly complete absorption into the systemic circulation.In Meyler's Side Effects of Drugs (Sixteenth Edition), 2016 Nervous system Thus, the aging process is associated with small changes in the kinetics of lorazepam. Absorption of IM and oral lorazepam was 80% to 100% complete. Peak plasma lorazepam concentrations were always reached within 2.5 hr, and values of absorption half-life (t1/2a) did not exceed 45 min. Deltoid IM injection and oral administration of tablets in the fasting state led to rapid absorption of lorazepam into the systemic circulation. 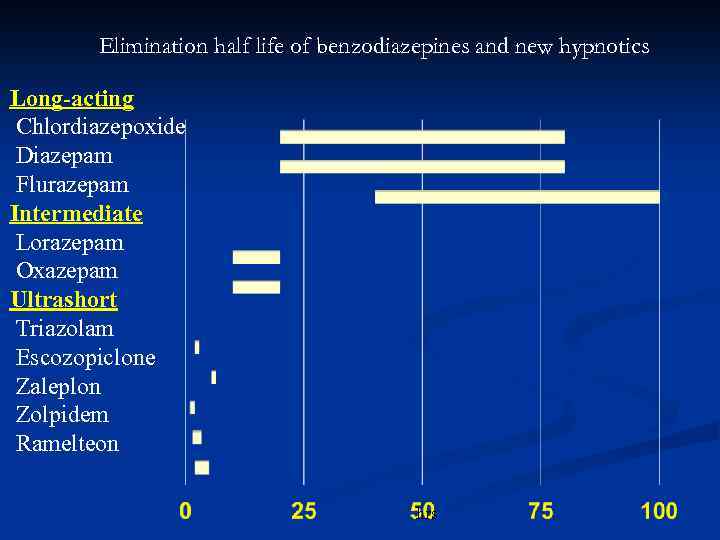
The rate and completeness of absorption of intramuscular (IM) and oral loraxepam was assessed in 10 of the elderly subjects. Gender had no apparent relationship to kinetics. Age differences in lorazepam clearance were partly explained by more frequent cigarette smoking in the young subjects. Values of elimination half-life (t1/2beta) in the elderly (15.9 hr) did not differ significantly from those in the young group (14.1 hr), but total clearance in the elderly (0.77 ml/min/kg) was 22% less (p less than 0.05) than in the young subjects (0.99 ml/min/kg). Volumes of distribution for lorazepam in the elderly group (mean, 0.99 1/kg), were slightly but significantly smaller than in the young group (1.11 1/kg), suggesting less extensive drug distribution in the elderly. The kinetic properties of single 1.5- to 3.0-mg doses of intravenous lorazepam were assessed in 15 healthy elderly subjects, 60 to 84 yr of age, and in 15 healthy young subjects, 19 to 38 yr of age. Lorazepam is a 3-hydroxy-1,4-benzodiazepine derivative biotransformed by glucuronide conjugation, followed by urinary excretion of the glucuronide metabolite.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
